|
Medical probiotics refer to live microorganisms that provide health benefits when consumed. Probiotics help maintain intestinal microbial balance and enhance digestion. They aid in treatment of various gastrointestinal disorders such as Irritable Bowel Syndrome, inflammatory bowel disease, and constipation. Growing prevalence of lifestyle diseases and gastrointestinal issues due to changing dietary patterns has increased demand for medical probiotics supplements globally. The global Medical Probiotics Market is estimated to be valued at US$ 4.43 Bn in 2024 and is expected to exhibit a CAGR of 19% over the forecast period 2024 to 2030, as highlighted in a new report published by Coherent Market Insights. Market Opportunity: The opportunity to improve gastrointestinal health is expected to drive the Medical Probiotics Market Demand. According to the report by the World Gastroenterology Organization, gastrointestinal disorders affect 60–70 million people in the US and 8–18% of the adult population worldwide each year. Probiotic supplements help balance the gut microbiota and promote a healthy intestinal environment, thereby aiding in treatment of gastrointestinal issues. They reduce risk of colon cancer, alleviate irritable bowel syndrome symptoms, and relieve constipation. Rising health concerns regarding gastrointestinal health will increase demand for medical probiotics for their multiple gut health benefits over the forecast period. Porter’s Analysis Threat of new entrants: The Medical Probiotics Market requires substantial R&D investments and manufacturing capabilities to produce high quality products. This makes it difficult for new companies to enter the market. Bargaining power of buyers: Individual consumers have low bargaining power as medical probiotics are generally prescribed by doctors for specific medical conditions. However, large healthcare institutions and bulk buyers can negotiate prices. Bargaining power of suppliers: The market has numerous raw material suppliers and is not dependent on a few major suppliers. This reduces supplier bargaining power. Threat of new substitutes: No viable alternatives currently exist for the health benefits provided by probiotic supplements in certain medical conditions. The threat from substitutes is low. Competitive rivalry: The market has numerous global and regional players. Intense competition on factors like product development, pricing and brand positioning is increasing rivalry among existing firms. SWOT Analysis Strengths: Probiotics help maintain intestinal microbial balance and support immune function. Clinical trials have demonstrated their efficacy in various gut disorders. Weaknesses: Standardization of strains and formulation across products. High R&D costs involved in developing new probiotic formulations for specific medical uses. Opportunities: Growing geriatric population worldwide prone to gut dysbiosis. Increasing adoption of probiotic supplements to complement antibiotic and IBD therapies. Scope for more clinical research on new disease applications. Threats: Stringent regulatory approval process for new strain/product approvals. Competition from substitute gut health products and microbiome-based therapies in the pipeline. Key Takeaways The global Medical Probiotics Market is expected to witness high growth at a CAGR of 19% between 2024 to 2031 driven by factors like rising gut health awareness, growing elderly population and prevalence of chronic digestive diseases. Regional analysis: North America currently dominates the Medical Probiotics Market led by the US. High healthcare spending, technological advancements and favorable regulatory policies support market growth. Asia Pacific is expected to be the fastest growing region during the forecast period led by countries like China, India and Japan. Rising living standards, large patient pools and growing health supplements industry are fueling medical probiotics demand in Asia. Key players: Key players operating in the Medical Probiotics Market are Alteryx, Inc., Bentley Systems Incorporated, ESRI, Furgo NV, General Electric, Hexagon AB, Google, Inc., MDA Corporation, Oracle Corporation, SAP SE, Trimble, Inc., and TOMTOM International, Inc. Major manufacturers are focusing on portfolio expansion through new product launches, acquisitions and partnerships to strengthen their market presence. Get More Insights on Medical Probiotics Market
0 Comments
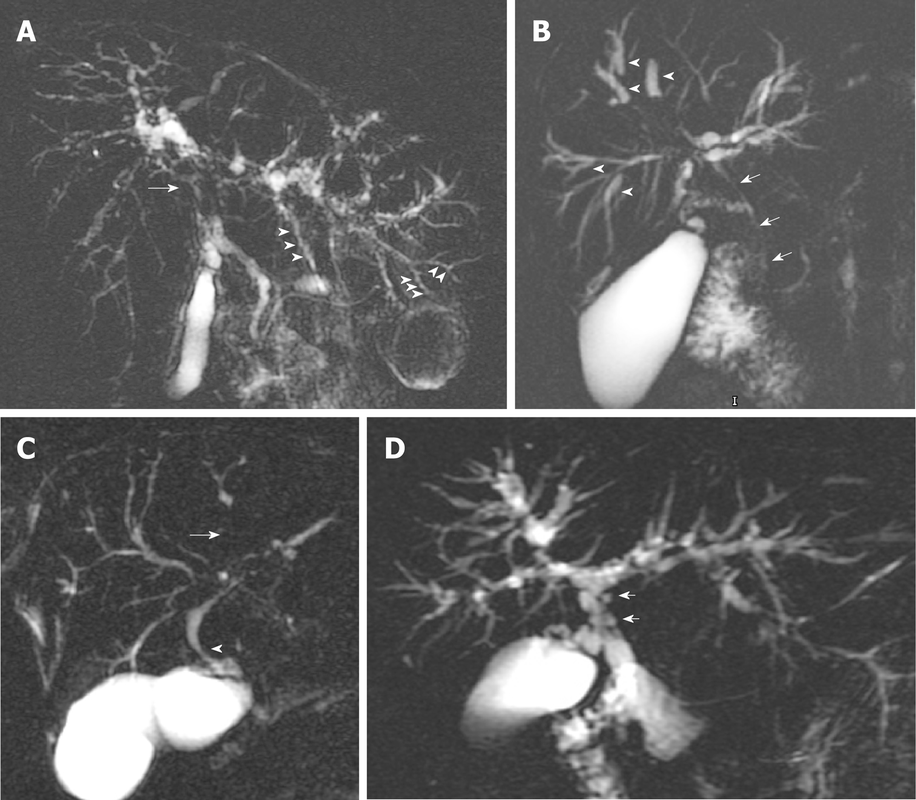
Nutraceutical excipients are ingredients used in the manufacturing of functional foods and dietary supplements to facilitate processing and improve product stability, texture, taste, appearance and shelf life. They help in creating an optimum physiochemical environment for the nutraceutical ingredients to act in the desired way. Some commonly used nutraceutical excipients include fillers, disintegrants, binders, coatings, preservatives, flavors and emulsifiers. Owing to the growing awareness about health and wellness, there has been significant demand for functional foods and dietary supplements in recent years. This has propelled the need for nutraceutical excipients to help manufacturers develop innovative formulations as per changing consumer preferences. The global Nutraceutical Excipient Market is estimated to be valued at US$ 3087.7 Bn in 2023 and is expected to exhibit a CAGR of 3.4% over the forecast period 2024 to 2031, as highlighted in a new report published by Coherent Market Insights. Market Opportunity: Increased Demand for Functional Food and Dietary Supplement Formulations The growing health consciousness and busy lifestyles have increased the demand for functional foods and dietary supplements that provide additional health benefits. Consumers are looking for products that can fulfill their daily nutrition intake as well as support specific health conditions. This presents a major market opportunity for nutraceutical excipient manufacturers. Functional foods containing nutraceutical excipients are in high demand for their ability to fortify staple foods with essential vitamins, minerals, proteins, fibers and phytochemicals. Similarly, dietary supplements require excipients to encapsulate or bind active ingredients into user-friendly delivery forms. The ability of excipients to enhance bioavailability, protect ingredients and modify release properties widens formulation possibilities for manufacturers. The burgeoning functional foods and supplements industry thus drives significant demand for innovative nutraceutical excipients. Porter’s Analysis Threat of new entrants: The nutraceutical excipient market has moderate threat of new entrants due to the presence of stringent regulations imposed on new entrants by several governments and higher initial costs involved in R&D. Bargaining power of buyers: There exists moderate bargaining power amongst buyers due to presence of a large number of manufacturers and differentiated product offerings. Bargaining power of suppliers: Suppliers have moderate bargaining power due to the availability of alternative raw material suppliers and commoditization of raw materials. Threat of new substitutes: Low threat of new substitutes emerging in the market as nutraceutical excipients have well-defined applications and regulation-defined inclusion limits. Competitive rivalry: High competitive rivalry exists amongst key players to strengthen their market presence through strategic expansions and new product launches. SWOT Analysis Strength: Flexible functionality of excipients, increasing adoption of nutraceuticals, growth in geriatric population. Weakness: High R&D costs, stringent regulations, vulnerability to raw material price fluctuations. Opportunity: Emerging markets offer high growth scope, technological advancements help widen applications. Threats: Alternative natural sources threaten demand, economic slowdowns reduce discretionary spending. Key Takeaways The global nutraceutical excipient market is expected to witness high growth over the forecast period supported by increasing consumer preference for healthy lifestyles. Global Nutraceutical Excipient Market Size is estimated to be valued at US$ 3087.7 Bn in 2023 and is expected to exhibit a CAGR of 3.4% over the forecast period 2024 to 2031. Regionally, North America dominates the market currently owing to rising health consciousness and spends on supplements in the US and Canada. Europe stands as the second largest market driven by growing elderly population demanding nutrition support. Key players operating in the nutraceutical excipient market are Arkema S.A., Evonik Industries A.G., Solvay S.A., Mitsubishi Gas Chemical Company Inc., Kemira Oyj, Thai Peroxide Limited (TPL), Taekwang Industrial Co. Ltd., PeroxyChem LLC, Hansol Chemical, and Nouryon. The regional analysis indicates Asia Pacific is anticipated to be the fastest growing region due to rising middle-class incomes and western influence on diets in major countries like India and China. Get More Insights on this Topic- https://www.newswirestats.com/nutraceutical-excipient-market-growth-insights-and-forecast/ Lidocaine patches are transdermal medicated adhesive patches that contain lidocaine which is an active ingredient that works by blocking sodium channels in the nerves and reducing the generation and transmission of pain signals to the brain. They provide localized pain relief without causing general sedation and are used to manage post-herpetic neuralgia, musculoskeletal pain and joint and back pain. Demand for effective and easy to use pain medication without the risk of opioid addiction is growing steadily which offers immense potential for lidocaine patches. The global Lidocaine Patches Market is estimated to be valued at US$ 6.47 Bn in 2024 and is expected to exhibit a CAGR of 10% over the forecast period 2024 to 2031, as highlighted in a new report published by Coherent Market Insights. Market Opportunity: The opportunity to provide effective pain relief without the risk of addiction and side effects of opioids is driving the growth of lidocaine patches market. They are convenient to use just by applying to the skin and provide localized relief to acute pain without causing sedation like oral pain medications. This reduces the risks associated with other medication forms. With growing aging population suffering from chronic pain conditions and rising rates of opioid addiction, demand for non-opioid alternative therapies is increasing. Lidocaine patches are emerging as a preferred treatment choice offering safe and easy to use pain relief. Manufacturers are anticipated to gain increasing market share by exploring this opportunity and expanding production capacity to cater to the rising global demand. Porter's Analysis Threat of new entrants: Low as significant capital is required for R&D and manufacturing. Established brands have advantage over new entrants. Bargaining power of buyers: High as presence of many private label brands increases competition. Buyers can negotiate for better prices and offers. Bargaining power of suppliers: Moderate as raw material suppliers have established relationships with manufacturers. Suppliers have limited influence over pricing. Threat of new substitutes: Low as lidocaine patches have few alternatives for localized pain relief. New products may pose threat if proven safer or more effective. Competitive rivalry: Intense as major players compete on pricing, product differentiation, advertising and promotion. Fierce competition to capture market share. SWOT Analysis Strengths: Established brands, product efficacy and safety profiles, extensive distribution networks. Weaknesses: Heavy R&D costs, stringent regulatory norms, vulnerability to price fluctuations of raw materials. Opportunities: Untapped markets in developing countries, scope for new product formulations, growing geriatric population drives demand. Threats: Patent expiries allow generic competition, social stigmas around opioids limit certain applications, reimbursement policies impact sales. Key Takeaways Global Lidocaine Patches Market Size is expected to witness high growth over the forecast period of 2024 to 2031.The market size for 2024 is estimated at US$ 6.47 Bn. North America dominates currently due to aggressive marketing by major players and rising sale of generics. However, Asia Pacific is emerging as the fastest growing regional market owing to large patient pool and increasing healthcare expenditure. Key players operating in the lidocaine patches market are Tilray, Canopy Growth Corporation, Aurora Cannabis, Aphria, Cronos Group, Acreage Holdings, MedMen Enterprises, Curaleaf Holdings, Trulieve Cannabis Corporation, and Organigram Holdings. The market is quite concentrated with top players dominating global sales. There have been no product approvals or major M&A activities recently. Get More insights on this topic- https://www.marketwebjournal.com/lidocaine-patches-market-value-analysis-and-forecast/ India and Oman pharmaceutical industry manufactures a wide range of generic drugs and medicines to cater to the diverse needs of patients across multiple therapeutic areas. The industry also provides cost-effective drugs which are highly accessible to people in both the countries owing to increasing generics penetration. India and Oman Pharmaceutical Industry Market is estimated to be valued at US$ 34 billion in 2024 and is expected to exhibit a CAGR of 9.3% over the forecast period 2024-2031, as highlighted in a new report published by Coherent Market Insights. Market Opportunity: The opportunity to meet rising healthcare expenditure in emerging economies presents a lucrative avenue for players in India and Oman pharmaceutical industry. Both India and Oman have seen rapid economic development over the past few decades which has increased disposable incomes and boosted healthcare spending. However, per capita healthcare expenditure remains relatively low in these countries compared to developed nations. This leaves significant headroom for growth as income and investment in healthcare infrastructure continue to increase. Local manufacturers are well-positioned to cater to this demand and expand access to affordable treatment options for a variety of ailments. By leveraging their low-cost production capabilities and expertise in generic drugs, industry players can gain a sizable share of the rising healthcare budgets in these emerging pharmaceutical markets. Porter’s Analysis Threat of new entrants: The pharmaceutical industry requires high capital investment in R&D for drug development which acts as a barrier for new companies. Regulatory compliances related to clinical trials and product approval further increases entry barriers. Bargaining power of buyers: The bargaining power of buyers is moderate due to the presence of generic manufacturers. However, customers have less influence over branded products which have strong brand loyalty. Bargaining power of suppliers: Large pharmaceutical companies have significant influence over API and raw material suppliers due to their large volume requirements and standards. Suppliers also face high switchig costs. Threat of new substitutes: Threat from new substitutes is low as drugs developed for different therapeutic areas require long development cycles and regulatory approvals. however, herbal and alternative medicine pose competition. Competitive rivalry: The industry has strong competition among existing players. Companies compete on basis of new drug launches, production quality, and expanding hospital and retail sales networks. SWOT Analysis Strength: Wide acceptance of allopathic treatment, large patient pool, growing healthcare spending. Emergence of CROs supporting clinical trials. Weakness: High dependency on imports for APIs and formulations, price controls affect margins. Low awareness in rural areas. Opportunity: Rising lifestyle diseases, growing geriatric population, scope for herbal drugs. Export potential to African and Asian markets. Threats: Stringent regulations delay approvals. Patent cliff of major drugs, price erosion due to generics. Key Takeaways India And Oman Pharmaceutical Industry Market Demand is expected to witness high growth during the forecast period owing to rising income levels, spreading health awareness, and government initiatives to strengthen healthcare infrastructure in both countries. Regionally, India dominates the market and holds the largest share currently. The country accounts for around 3% of the global pharmaceutical production by volume. Favourable regulations, skilled manpower and presence of leading facilities are driving India's leading position. However, Oman is poised to be the fastest growing market in the region backed by initiatives to develop local manufacturing capabilities and reduce import dependence for drugs and medical devices. Key players operating in India and Oman pharmaceutical industry are Sun Pharmaceutical Industries, Cipla, Dr. Reddy's Laboratories, AstraZeneca and GlaxoSmithKline. The market is quite competitive with the top 5 companies accounting for over 40% share. Companies are focused on expanding product portfolios through collaborations and leveraging theirstrong distribution network across various regions. However,smaller domestic players also have a growing presence, intensifying the competitive landscape. Get More Insights on this Topic- https://www.dailyprbulletin.com/india-and-oman-pharmaceutical-industry-market-growth-analysis/ Genomic cancer testing involves analyzing the complete set of genes or genome of cancerous cells or a tumor to check for any mutations or other changes that can help determine a patient's risk for cancer, detect cancer in early stages, prognosis, targeted therapies, and monitor recurrence. Such testing offers oncologists and physicians deep clinical insights to develop more personalized and targeted treatment plans for cancer patients. The global Genomic Cancer Testing Market is estimated to be valued at US$ 14.66 billion in 2023 and is expected to exhibit a CAGR of 7.8% over the forecast period 2023-2030, as highlighted in a new report published by Coherent Market Insights. Market Opportunity: The opportunity to drive personalized medicine is expected to boost the genomic cancer testing market during the forecast period. Genomic cancer testing provides vital insights about specific mutations in a patient's tumor which help physicians customize treatment plans. For example, genomic profiling can identify patients that are likely to respond to certain targeted therapies through analysis of specific gene mutations. This enables administration of most effective drugs and avoids exposure to ineffective or harmful treatments. Personalized treatment improves clinical outcomes and survival rates of cancer patients. With continuous advancements in precision oncology, genomic cancer testing is poised to play a pivotal role in driving wider adoption of personalized medicine approaches in cancer treatment over the coming years. Porter’s Analysis Threat of new entrants: The threat of new entrants in the genomic cancer testing market is low as it requires huge capital investments to set up facilities for conducting genetic tests and analysis. It also requires professional expertise in oncology and genetics which make the entry barriers high for new players. Bargaining power of buyers: The bargaining power of buyers is moderate in the genomic cancer testing market. The test results directly impact the treatment decisions, so buyers have moderate control over price. However, multiple service providers are available. Bargaining power of suppliers: The bargaining power of suppliers is low in the genomic cancer testing market due to the availability of substitute raw material and technology suppliers. Suppliers do not have pricing leverage. Threat of new substitutes: The threat of new substitutes is low as genomic cancer testing provides precise diagnosis and personalized treatment which cannot be substituted. Competitive rivalry: The competitive rivalry is high as the major players actively compete on the basis of pricing, innovation, and service quality. SWOT Analysis Strength: Availability of advanced technologies for comprehensive genomic profiling and analysis. Growing demand for personalized cancer treatment. Weakness: High cost of tests limits widespread adoption. Requirement of well-equipped labs and skilled professionals. Opportunity: Increasing incidence of cancer worldwide. Growing healthcare expenditure in emerging economies. Threats: Stringent regulations for product approval. Reimbursement issues especially in developing nations. Key Takeaways Global Genomic Cancer Testing Market Size is expected to witness high growth during the forecast period of 2023-2030. Regional analysis: The North America genomic cancer testing market is expected to continue its dominance during the forecast period due to increasing incidence of cancer, favorable reimbursement policies, and adoption of advanced cancer diagnostic tools. The Asia Pacific market is projected to witness the highest growth rate owing to rising healthcare expenditure, improving healthcare infrastructure, and growing medical tourism industry in the region. Key players operating in the genomic cancer testing market are Myriad Genetics, Illumina, Roche, Qiagen, Thermo Fisher Scientific, among others. Myriad Genetics offers a comprehensive portfolio of tests for hereditary cancer risk assessment, including BRACAnalysis, BRACAnalysis CDx, Colaris, and Colaris AP. Get More Insights on this Topic https://www.rapidwebwire.com/genomic-cancer-testing-market-forecast-outlook-and-growth/ The opioid use disorder market involves medical products and therapies used to treat addiction and manage withdrawal symptoms associated with opioid abuse. Some key products in this market include buprenorphine, naltrexone, methadone, and various abuse-deterrent opioid formulations. The demand for these drugs and therapies has grown in recent years as the prevalence of opioid abuse and addiction has reached epidemic levels in various countries. The global opioid use disorder market is estimated to be valued at US$ 3236.55 Mn in 2023 and is expected to exhibit a CAGR of 8.0% over the forecast period 2023 to 2030, as highlighted in a new report published by Coherent Market Insights. Market Opportunity: The increased availability of more effective and long-acting treatment options presents a major market opportunity in the opioid use disorder market. Drugs like extended-release buprenorphine and six-month implant formulations help address relapse and non-adherence concerns associated with traditional treatment regimens. Their ability to provide lasting suppression of withdrawal symptoms and drug cravings can help more patients stay in remission long-term. This in turn can boost recurrence rates and support the continued scaling up of opioid addiction treatment services worldwide. Porter’s Analysis Threat of new entrants: The opioid use disorder market requires immense capital investments for Research & Development of new therapies. Stringent regulations in the pharmaceutical industry impose additional costs that deter new players. Bargaining power of buyers: Individual opioid use disorder patients have limited influence over pricing and product offerings. However, pharmaceutical companies face pricing pressure from large healthcare providers and insurance companies. Bargaining power of suppliers: The market involves few raw material suppliers and vendors of medical devices and treatment equipment have some influence over API and components pricing. This grants suppliers modest bargaining power. Threat of new substitutes: Alternatives like psychotherapy and behavioral therapies pose a threat. However, they are not always as effective as medication therapy which limits their usage as perfect substitutes. Competitive rivalry: The market involves many global players and generics manufacturers. Players engage in intensive efforts to develop novel therapies via collaborations and acquisitions to gain competitive advantages. SWOT Analysis Strengths: Growing awareness, strong R&D pipelines, increasing accessibility of treatments. Weaknesses: Social stigma, risk of relapse, complex pathology necessitating multi-modal therapies. Opportunities: Untapped emerging markets, scope for combination therapies, digital therapeutics. Threats: Patent expiries of blockbuster drugs, pricing pressures, inconsistencies in regulations. Key Takeaways Global Opioid Use Disorder Market Demand is expected to witness high growth over the forecast period owing to rising prescription drug abuse and need for effective treatment options. Regional analysis of the North America opioid use disorder market comprises the US and Canada. The US holds the lion’s share owing to growing public health crisis. According to CDC, opioid overdose claims over 100 American lives daily in the US. This has spurred government bodies and private players to boost funding for advanced therapeutics. Canada follows US trends and is working towards resolving inconsistencies in provincial policies governing opioid prescriptions and treatment access. Key players operating in the opioid use disorder market are Moog Inc., Honeywell International Inc., AMETEK Inc., TE Connectivity, Woodward Inc., General Dynamics Corporation, Parker Hannifin Corp, Sensata Technologies, Tamagawa Seiki Co., Ltd., LTN Servotechnik GmbH. Major companies are focusing on launching novel MAT options such as buprenorphine and methadone drugs, extended-release injectables, as well as digital therapeutics to tap the high market potential. Personalized genomics refers to genetic testing services that analyze a person's entire genome or parts of their genome to understand their health risks, ancestry composition or other traits. These services allow consumers to gain important biological insights from direct-to-consumer genetic tests. Products in this space include DNA collection kits, raw DNA data reports, personalized health reports and ancestry information. The global personalized genomics market is estimated to be valued at US$ 9.14 Bn in 2023 and is expected to exhibit a CAGR of 0.0% over the forecast period 2023 to 2030, as highlighted in a new report published by Coherent Market Insights. Market Dynamics: One of the key drivers for the high growth of the personalized genomics market is the rapid advancements in genome sequencing technologies over the past decade. Next generation sequencing platforms have made whole genome sequencing more affordable and accessible. Technological breakthroughs such as long read sequencing allow for better structural variation detection. Increased computing power also enables easier analysis and interpretation of large genomic datasets. Additionally, declining costs of sequencing a whole human genome from over $3 million in 2007 to under $600 now is fueling widespread adoption of personalized genomic services. The commercialization of direct-to-consumer genetic tests also allows more people to access their genomic insights. Furthermore, growing awareness about the health benefits of genetic screening is widening the customer base for personalized genomics globally. SWOT Analysis Strength: The personalized genomics market has seen significant advancements in sequencing technologies and genome analysis over the years. Genome sequencing costs have declined significantly, making the tests more affordable. There is growing awareness about the importance of personalized healthcare and genetics which is driving demand. Weakness: However, the regulatory landscape for these tests is still evolving. Ethical and privacy issues surrounding genetic data are a concern. There are also questions around the clinical utility and limitations of some direct-to-consumer genetic tests available without physician involvement. Opportunity: Developing nations and emerging economies represent a major market opportunity as their healthcare systems expand and individuals can afford these tests. New applications around diet, ancestry, drug response and more based on genetic insights can further propel demand. Direct collaborations with healthcare providers bring new customers. Threats: Established players face competition from new entrants offering novel approaches. Strict regulations in some countries also pose market access challenges. Potential security breaches of sensitive genetic data stored by companies could erode customer trust over time. Key Takeaways The global personalized genomics market is expected to witness high growth over the forecast period of 2023 to 2030. Global Personalized Genomics Market Size is estimated to be valued at US$ 9.14 Bn in 2023 and is expected to exhibit a CAGR of 0.0% over the forecast period 2023 to 2030. Regional analysis shows that North America currently dominates due to advanced healthcare systems and high acceptance rates. However, Asia Pacific is poised to be the fastest growing market led by countries like China, India and Japan with their large populations and improving medical infrastructures. Key players operating in the personalized genomics market are 23andMe, Ancestry, Mapmygenome, Helix, MyHeritage, Gene by Gene, Pathway Genomics, Veritas Genetics, Xcode, Color Genomics, Positive Biosciences, Futura Genetics, Myriad Genetics, Quest Diagnostics, Laboratory Corporation of America, Illumina, Thermo Fisher Scientific, Qiagen, Genomic Health and Nebula Genomics. 23andMe and Ancestry are currently the leaders in the direct-to-consumer genetic testing space while Illumina, Thermo Fisher, Qiagen and others provide instruments, kits, services and platforms for research applications and clinical use. Get More Insights on this Topic- https://www.marketwebjournal.com/personalized-genomics-market-size-share-and-demand/ PARP inhibitors are a class of anti-cancer drugs that help treat cancers caused by mutations in BRCA1 and BRCA2 genes, like breast and ovarian cancers. They work by blocking an enzyme called poly (ADP-ribose) polymerase that helps repair damaged DNA, thereby allowing chemotherapy or radiation to kill cancer cells more effectively. With the growing cases of breast and ovarian cancers, the demand for PARP inhibitors as an important line of treatment for BRCA mutation patients is increasing tremendously. The global PARP inhibitor Market is estimated to be valued at US$ 6.13 Bn in 2023 and is expected to exhibit a CAGR of 6.8% over the forecast period 2023 to 2030, as highlighted in a new report published by Coherent Market Insights. Market Dynamics Increasing adoption in oncology treatment: The key driver for the growth of the PARP inhibitor market is its increasing adoption in the treatment of various cancers like breast and ovarian cancers. As per the National Cancer Institute, PARP inhibitors have shown promising results in slowing or stopping the growth of cancer cells with BRCA1 or BRCA2 gene mutations. This is significantly driving the demand for PARP inhibitors in oncology treatment. Another major driver is the rising number of approvals for new PARP inhibitor drugs to treat additional cancer types. For example, Lynparza by AstraZeneca has been approved to treat pancreatic cancer and prostate cancer with BRCA mutations in recent times, thereby expanding its eligible patient pool. Such additional approvals are supporting the market growth of PARP inhibitors. SWOT Analysis Strength: PARP inhibitors have shown promising results in clinical trials for several cancers with DNA repair deficiencies like BRCA mutations. Three large pharmaceutical companies have obtained FDA approval for their PARP inhibitor drugs to treat ovarian and breast cancers in recent years, gaining first mover advantage in the market. Weakness: The high cost of PARP inhibitor drugs pose an accessibility challenge for many cancer patients. Adverse effects like fatigue and anemia have also been reported, requiring close patient monitoring. Truncated treatment durations also reduce the potential revenues for pharmaceutical companies. Opportunity: Ongoing research continues to explore new potential indications for PARP inhibitors in prostate, pancreatic and other solid tumor types. Combination therapies with chemotherapy, immunotherapy or radiation are being studied to improve efficacy. Expanding insurance coverage and pricing strategies could help address the cost concerns over time. Threats: Competition from cheaper generics once initial patents expire could erode market share of leading branded PARP inhibitors. Failure in late stage clinical trials for new indications would also hamper revenue growth projections for companies. Emergence of alternative targeted therapies may reduce the addressable patient pool eligible for PARP inhibitors. Key Takeaways Global PARP Inhibitor Market Trends are expected to witness high growth over the forecast period driven by continued approvals for additional cancer indications. With several large pharmaceutical companies engaged in ongoing clinical research, the therapeutic potential of PARP inhibitors is yet to be fully realized. Regional analysis: North America currently dominates the PARP inhibitor market owing to early drug approvals and availability of specialized cancer care centers. However, the Asia Pacific region is expected to see the fastest gains through the forecast period due to rising healthcare investments, growing cancer burdens and increasing adoption of novel targeted therapies in countries like China, India and Japan. Key players: Key players operating in the PARP inhibitor market are FLSmidth & Co. A/S, Metso Oyj, Koppern Group, CITIC Heavy Industries Co. Ltd. (CITIC HIC), ThyssenKrupp Industrial Solutions AG (Polysius AG), SGS S.A., KHD Humboldt Wedag International AG, Outotec Oyj, and ABB Ltd. These companies have made substantial R&D investments to explore additional indications and combination regimens to maximize the clinical utility of PARP inhibitors. Get More Insights on this Topic- https://www.marketwebjournal.com/parp-inhibitor-market-size-share-and-outlook/ Ophthalmic drugs are medications that are applied topically to the eyes to treat various eye diseases and disorders. Common ophthalmic drugs include artificial tears to treat dry eyes, antibiotics and anti-fungal drops to treat eye infections, anti-glaucoma drugs to reduce pressure in the eyes, and anti-inflammatory drugs to treat eye conditions associated with redness and swelling. The Global Ophthalmic Drugs Market is estimated to be valued at US$ 48.19 Bn in 2023 and is expected to exhibit a CAGR of 8.9% over the forecast period 2023 to 2030, as highlighted in a new report published by Coherent Market Insights. Market Dynamics: The growth of the ophthalmic drugs market is majorly driven by the increasing prevalence of various eye diseases globally. As per credible reports, around 2.2 billion people suffer from vision impairment or blindness across the world. Age-related macular degeneration, diabetic retinopathy, glaucoma, and cataract are some of the major causes of blindness. Thus, the rising cases of such eye disorders have augmented the demand for effective treatment options like ophthalmic drugs. Furthermore, the rapid technological advancements in drug delivery systems like eye drops, gels, and ointments have provided improved drug absorption and bioavailability, thereby boosting market growth. The other driver for the market growth is the increasing geriatric population worldwide who are more prone to developing age-related eye conditions. According to the World Health Organization, the number of people aged 65 years and older is expected to rise from an estimated 524 million in 2010 to nearly 1.5 billion in 2050. The aged population is more at risk of eye problems like glaucoma, cataracts, diabetic retinopathy, and age-related macular degeneration, thereby propelling the demand for ophthalmic drugs. SWOT Analysis Strength: The ophthalmic drugs market has seen strong growth in the development of new drugs to treat eye conditions. Several major drug companies have large pipelines of drugs in clinical trials that aim to treat glaucoma, macular degeneration and other diseases. Additionally, the aging global population has led to a higher prevalence of eye conditions that require medication treatment. Weakness: Development of new drugs requires significant investments of both time and capital from pharmaceutical companies. Many drug candidates fail during clinical trials, representing billions of dollars in wasted research costs. Additionally, generic competition erodes sales when patents expire on blockbuster drugs. Opportunity: Asia Pacific and Latin America regions provide opportunities for market growth as health standards and spending power rise in emerging economies. New drug delivery methods such as sustained release gels and implants also provide opportunities for innovator companies to extend patent protection of existing drugs. Threats: Expiry of drug patents leads to competition from inexpensive generic versions, causing a decline in revenues for innovator companies. Stringent regulations from authorities can also delay drug approvals and market entry. Key Takeaways Global Ophthalmic Drugs Market Demand is expected to witness high growth. The Global Ophthalmic Drugs Market is estimated to be valued at US$ 48.19 Bn in 2023 and is expected to exhibit a CAGR of 8.9% over the forecast period 2023 to 2030. Asia Pacific region is projected to be the fastest growing market during the forecast period due to rising healthcare expenditure and increasing demand for ophthalmic drugs. Key players related content comprises Key players operating in the ophthalmic drugs market are IBM, Cisco, Check Point, Trellix, Trend Micro, NortonLifeLock, Rapid7, Micro Focus, Microsoft, Amazon Web Services, Oracle, Fortinet, Palo Alto Networks, Accenture, CyberArk, SentinelOne, Qualys, F-Secure, F5, DataVisor, RevBits, WiJungle, BluVector, Aristi Labs, Imperva, Securden, Forcepoint, Sophos, RSA Security, Proofpoint, Juniper Networks, Splunk, SonicWall, Algosec, Zscaler, Cynet, and Nozomi Networks. Get More Insights on this Topic- https://www.marketwebjournal.com/ophthalmic-drugs-market-demand-value-and-forecast/ Primary sclerosing cholangitis (PSC) is a chronic progressive cholestatic liver disease characterized by inflammation and fibrosis of both intrahepatic and extrahepatic bile ducts. PSC occurs in patients with ulcerative colitis and is associated with an increased risk of cholangiocarcinoma and colon cancer. The main symptoms associated with PSC include fatigue, itching, abdominal pain, and clay-colored stools. PSC is currently diagnosed through a magnetic resonance cholangiopancreatography (MRCP) scan or endoscopic retrograde cholangiopancreatography (ERCP) which help identify irregularities and stricturing of the bile ducts. Existing treatment options include ursodeoxycholic acid (UDCA) to improve liver function and bile flow along with management of symptoms through antipruritics, antibiotics, and bile acid sequestrants. Promising pipeline drugs target key pathways involved in inflammation, fibrosis, and bile acid metabolism with the aim to delay disease progression and reduce complications. The global Primary Sclerosing Cholangitis market is estimated to be valued at US$ 1.98 Mn in 2023 and is expected to exhibit a CAGR of 5.5% over the forecast period 2023 to 2030, as highlighted in a new report published by Coherent Market Insights. Market Dynamics: One of the major drivers fueling the growth of the Primary Sclerosing Cholangitis Market is the promising pipeline drugs in late-stage clinical trials targeting various disease mechanisms. Some of the key pipeline candidates include bezafibrate, cenicriviroc, and obeticholic acid. Bezafibrate, a pan-PPAR agonist, is presently in Phase 3 clinical trial to evaluate its efficacy in improving biochemical markers and delaying histological progression in PSC patients. Cenicriviroc, a dual CCR2 and CCR5 antagonist, is in Phase 2b clinical trial for targeting immune cells implicated in cholangitis and fibrosis. Obeticholic acid, a farnesoid X receptor agonist, recently completed Phase 3 clinical trial and showed ability to reduce alkaline phosphatase levels and improve liver stiffness. If approved, these novel drugs have potential to provide better treatment outcomes than existing options SWOT Analysis Strength: Primary sclerosing cholangitis (PSC) has diagnostic tests that help confirm a diagnosis such as magnetic resonance cholangiopancreatography (MRCP) which produces detailed images of the bile ducts and pancreatic ducts without using radiation. Endoscopic retrograde cholangiopancreatography (ERCP) also helps in diagnosing PSC by examining the bile ducts and detects abnormalities. Blood tests can detect elevated levels of liver enzymes indicating potential liver damage. Weakness: There is no known cure for PSC and existing treatment options only help manage symptoms and slow disease progression. Liver transplantation may be the only treatment option for patients with end stage liver diseases. Recurrence of PSC in the transplanted liver is also a concern. Risk of developing colorectal cancer is also a weakness as nearly one third of PSC patients develop it. Opportunity: Development of new drug therapies targeting specific pathways involved in PSC can help treat the underlying disease process. Combination drug therapies involving immunomodulator drugs, anti-fibrotics and immunosuppressants hold promise. Advances in surgical techniques and post transplantation care can increase eligibility for and success of liver transplantation in PSC patients. Threats: Symptoms of PSC often overlap with other liver conditions making diagnosis difficult. Compliance to lifelong medications is a challenge post transplantation. Risk of surgical complications and organ rejection during and after transplantation pose threats. Key Takeaways Global Primary Sclerosing Cholangitis Market Demand is expected to witness high growth. The market size is projected to increase from US$ 1.98 million in 2023 to US$ 3.11 million by 2030, growing at a CAGR of 5.5% during the forecast period. Regional analysis: North America dominates the global PSC market currently owing to increasing prevalence of IBD and rising healthcare expenditure. The region accounts for the largest market share currently and is expected to maintain its dominance over the forecast period. Europe is the second largest market for PSC currently. Key players: Key players operating in the primary sclerosing cholangitis market are Abbott Point of Care Inc., Cornley Hi-tech Co., LTD, Radiometer Medical ApS, EDAN Instruments, Inc., ERBA Diagnostics Mannheim GmbH, F Hoffman La Roche Ltd., Fortress Diagnostics, Instrumentation Laboratories, LifeHealth, Medica Corporation, Nova Biomedical Corporation, OPTI Medical Systems, Inc., Sensa Core, Siemens Medical Solutions USA, Inc. Get More Insights on this Topic- https://www.rapidwebwire.com/primary-sclerosing-cholangitis-market-demand-size-and-share/ Check below trending articles related to this topic- https://careersplay.com/the-rising-trend-of-biohacking-and-its-opportunities |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
April 2024
Categories
All
|





 RSS Feed
RSS Feed
